Postgraduate Seminar: Peter Canfield
Wednesday, 7 October 2020. 11am – 12pm.
This seminar will be delivered via Zoom – Please email chemistry.researchsupport@sydney.edu.au for zoom link and password.
Peter Canfield, PhD Candidate, School of Chemistry.
Host: Emeritus Prof. Maxwell Crossley
Akamptisomerisation: the final fundamental type of stereoisomerism and how it shows the way forward to the complete conceptualisation of Chemical Space.
The experimental demonstration, in a B(F)-O-B(F) coordinated porphyrin, of isolable stereoisomers that are related only by “angle inversion” of the bent B-O-B centre proved to be a previously unclassified and final fundamental type of stereoisomerism that we call akamptisomerism.
In the process of classifying akamptisomerism, it was discovered that this type of stereoisomerism is not restricted to obscure molecules of only academic interest but is widely prevalent with applications to Medicinal Chemistry, natural products, and Materials Science.
Further, and of greatest impact, this discovery has revealed deficiencies in the current ad hoc IUPAC practices and leads to the suggestion of a systematic approach for new terminology and the conceptualisation of all Chemical Space empowering both human and machine led research.
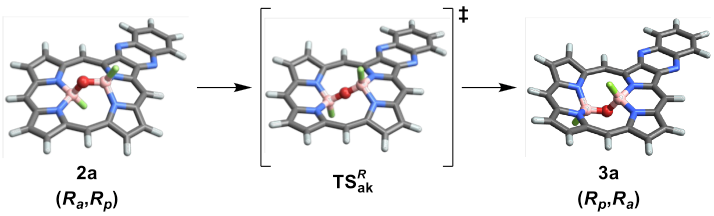
Canfield, P.J., Blake, I.M., Cai, Z. et al. A new fundamental type of conformational isomerism. Nature Chem 10, 615–624 (2018). https://doi.org/10.1038/s41557-018-0043-6

